MaaT013 (Xervyteg®): Positive Results from the Pivotal Study in Acute
Graft-vs-Host Disease
Study Design
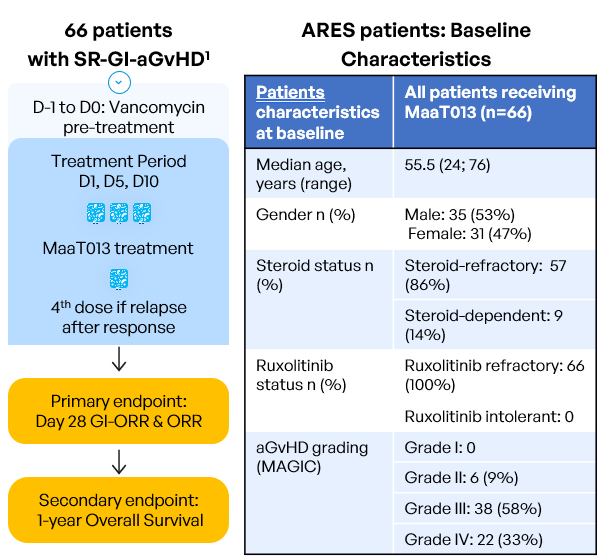
Inclusion criteria:
- Age ≥ 18 years old
- Allo-HSCT with any type of donor, stem cell source, GvHD prophylaxis or conditioning regimen
- Patients who develop aGvHD episode with GI involvement per MAGIC guidelines (=grades II to IV), with or without involvement of other organs (Harris et al. 2016)
- Patients resistant to steroids AND either resistant to OR with intolerance to ruxolitinib (intolerant patients: who had grade 3 or higher treatment-emergent and ruxolitinib-attributed adverse event that did not resolve within 7 days of discontinuing ruxolitinib). The diagnosis must be confirmed within 48h prior to study pre-treatment start.
The study was conducted in 6 European countries (Austria, Belgium, France,Germany, Italy and Spain) accross 50 sites.
ARES: Strong Response to MaaT013 (Xervyteg®) in aGvHD Following Steroid and Ruxolitinib Failure
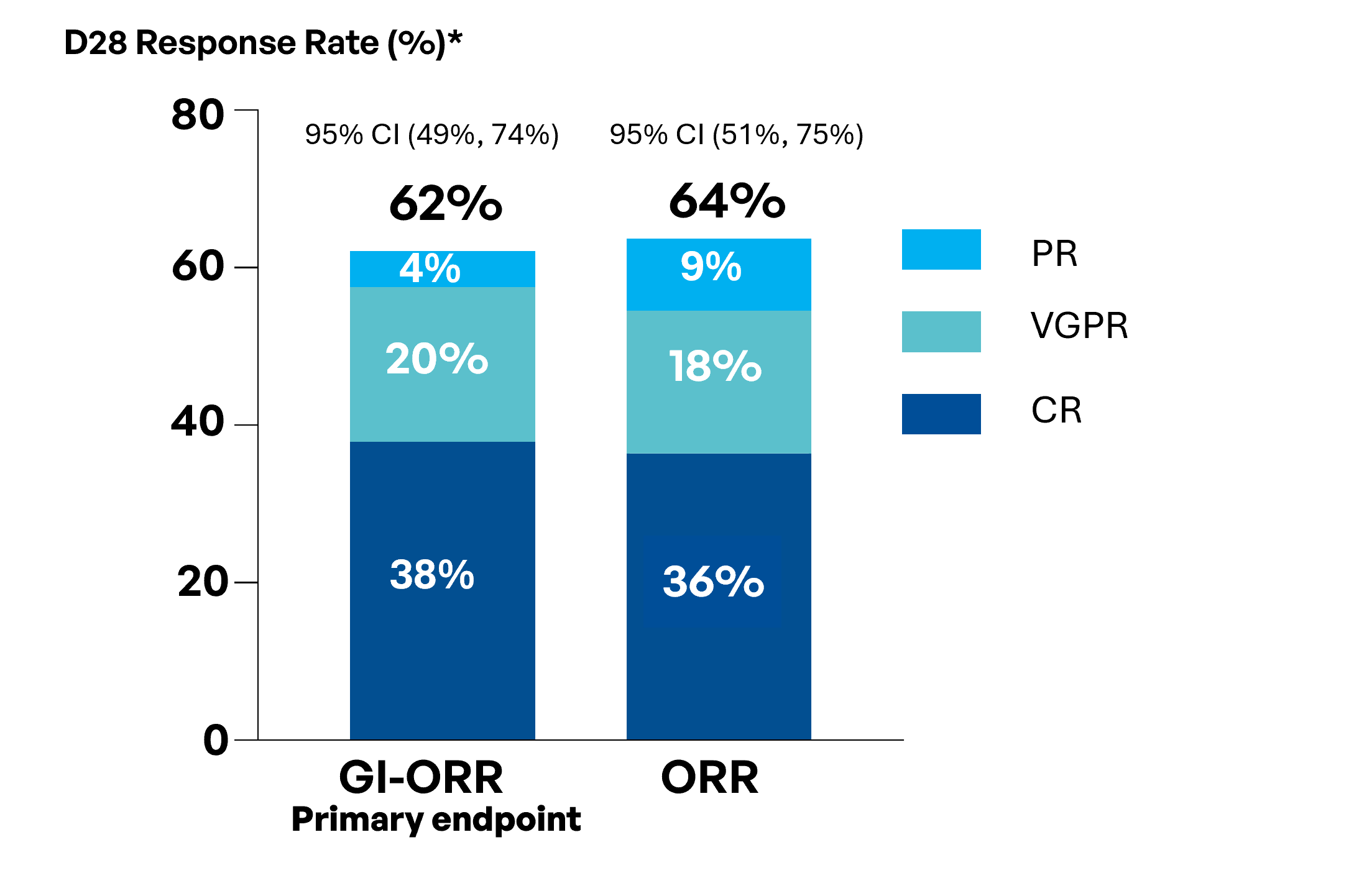
62% GI-ORR with high CR and VGPR rates.
64% ORR indicating efficacy beyond the GI tract.
- Day 56: Durable efficacy with GI-ORR 47% and all-organ ORR 45%.
- Month 3: GI-ORR and all-organ ORR both at 44%, confirming sustained response.
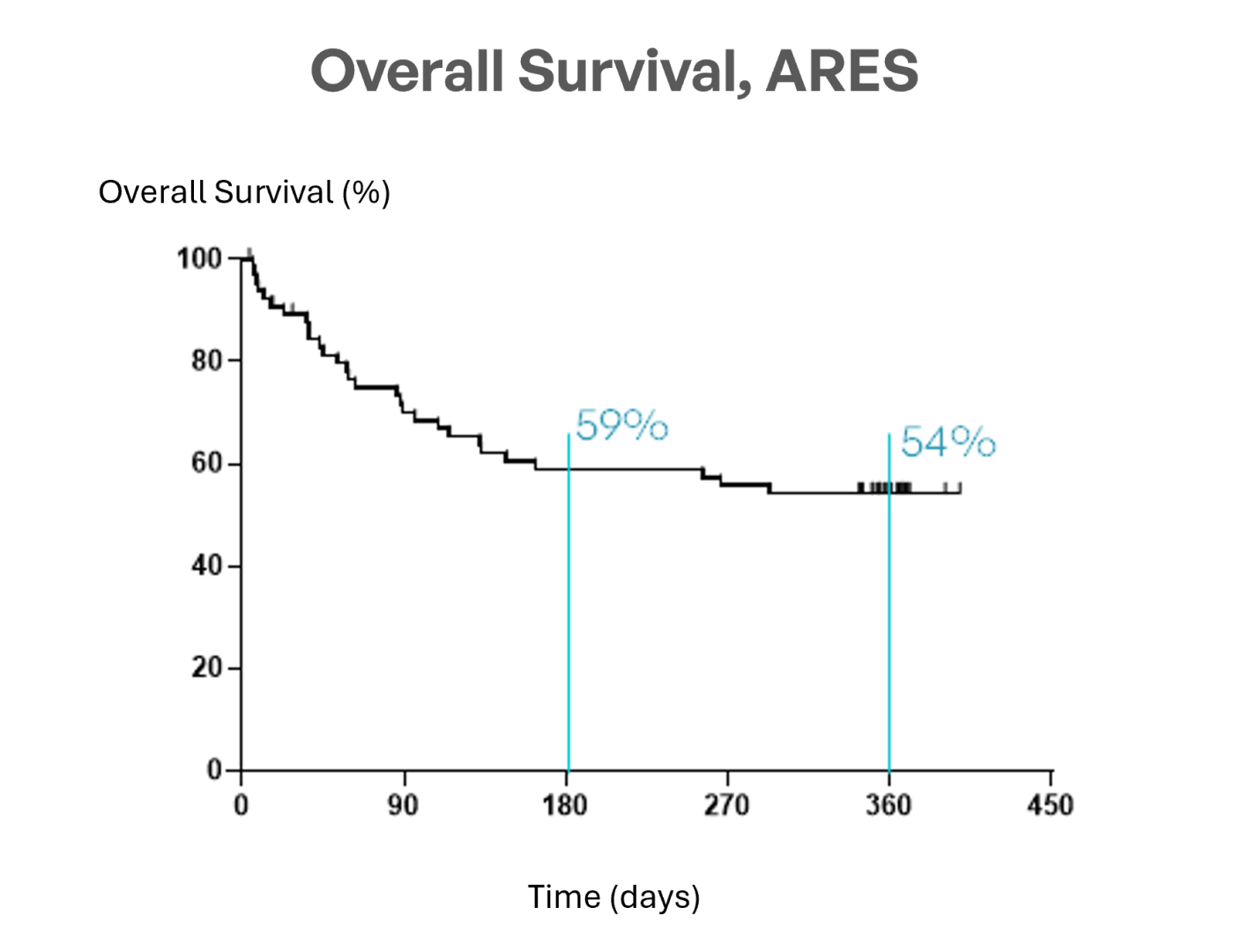
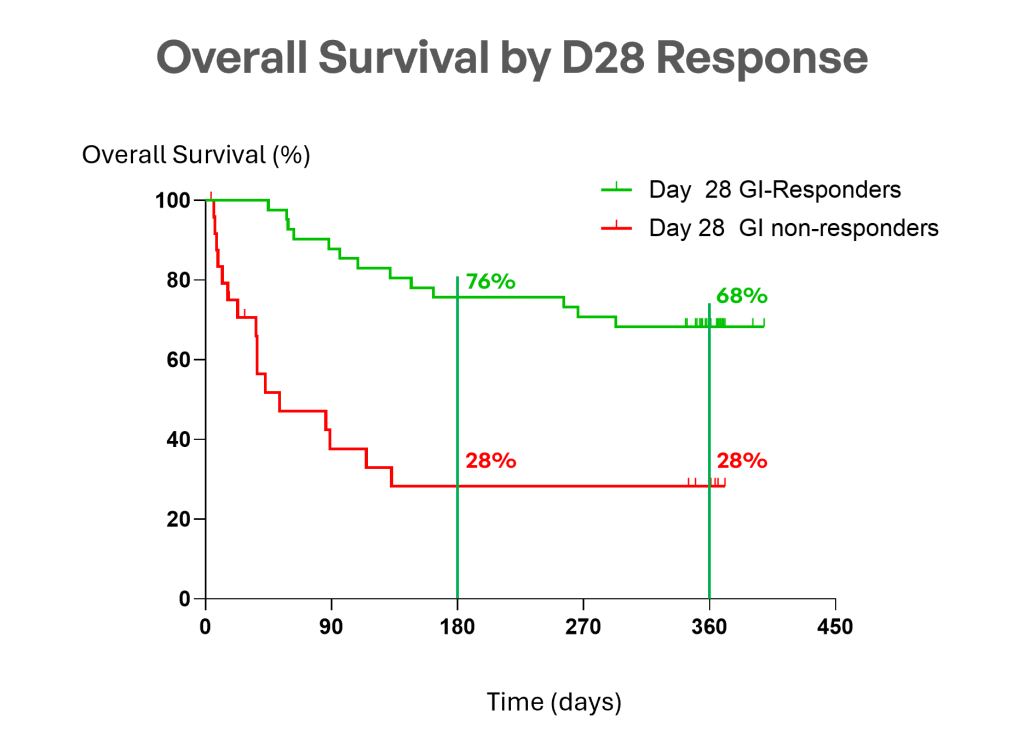
MaaT013 (Xervyteg®) achieves a 54% 1‑year OS, outperforming expectations, whereas the contemporaneous European CHRONOS1 cohort with ARES overlapping centers and inclusion aligned criteria reported 29% survival at EBMT Congress 2026 and published in BMT journal, with retrospective design considerations.
1: 3rd-line with aligned eligibility. Non-randomized, non-matched; descriptive only – CHRONOS STUDY – Clausen et al., BMT 2026
“These results confirm that MaaT013 offers a durable clinical benefit for patients with GI-aGvHD. Achieving a 62% GI-ORR at Day 28, maintaining responses over time, and reaching a 54% one-year overall survival represent a meaningful step forward in addressing this critical unmet need.”
Prof. Malard, MD, hematology professor at Saint-Antoine Hospital and Sorbonne University, lead investigator for the ARES pivotal trial

📰 MaaT013 (Xervyteg®) is currently under evaluation by the European Medicines Agency (EMA) following the submission of a Marketing Authorization Application in June, for the treatment of adult patients with aGvHD, including gastrointestinal involvement, who have received two prior lines of therapy.
- 1SR-GI-aGvHD in ARES Study is defined as patients resistant to steroids AND either resistant to OR with intolerance to ruxolitinib (intolerant patients: who had grade 3 or higher treatment-emergent and ruxolitinib-attributed adverse event that did not resolve within 7 days of discontinuing ruxolitinib). SR-aGvHD includes patients administered high-dose systemic CS (methylprednisolone 2 mg/kg/day – or equivalent prednisone dose 2.5 mg/kg/day), given alone or combined with CNI or mTOR inhibitor and either:
- Lack of improvement (i.e., no decrease in stage in at least 1 involved organ system) after ≥ 5 days of treatment with CS at 2 mg/Kg/d methylprednisolone equivalent dose, or
- Progression (i.e., increase in any organ system or any new organ involvement) after ≥ 3 days of treatment with CS at 2 mg/Kg/d methylprednisolone equivalent dose, or
- Patients treated with 1 mg/Kg/d of CS because the physician deemed that they would not tolerate 2 mg/Kg/d and who correspond to the definition of SR patients, or
- Patients who previously began CS therapy at a lower dose (at least 1 mg/Kg/d methylprednisolone equivalent) but develop new GvHD in another organ system, or
- Patients who cannot tolerate CS tapering, i.e., begin CS at 2.0 mg/Kg/d, demonstrate response, but show disease progress before a 50% decrease from the initial starting dose of CS is achieved.
- Resistance to ruxolitinib is defined as any of the following (Mohty M. 2020):
- Progression of GvHD compared to baseline after at least 5 days of treatment with ruxolitinib, based either on objective increase in stage/grade, or new organ involvement, or
- Lack of improvement in GvHD (partial response or better) compared to baseline after at least 14 days of treatment with ruxolitinib, or
- Loss of response, defined as objective worsening of GvHD determined by increase in stage, grade or new organ involvement at any time after initial improvement, or
- Absence of complete response or very good partial response at day 28 after ruxolitinib.
- Intolerance to ruxolitinib is defined as GvHD manifestations persisting without improvement in patients who had grade 3 or higher treatment-emergent and ruxolitinib-attributed adverse event that did not resolve within 7 days of discontinuing ruxolitinib.
- aGVHD: acute graft-versus-host disease
- GI:gastrointestinal
- MAGIC: Mount Sinai Acute GVHD International Consortium
- ORR: overall response rate
- SR: steroid-refractory
- D: Day
- M: Month

