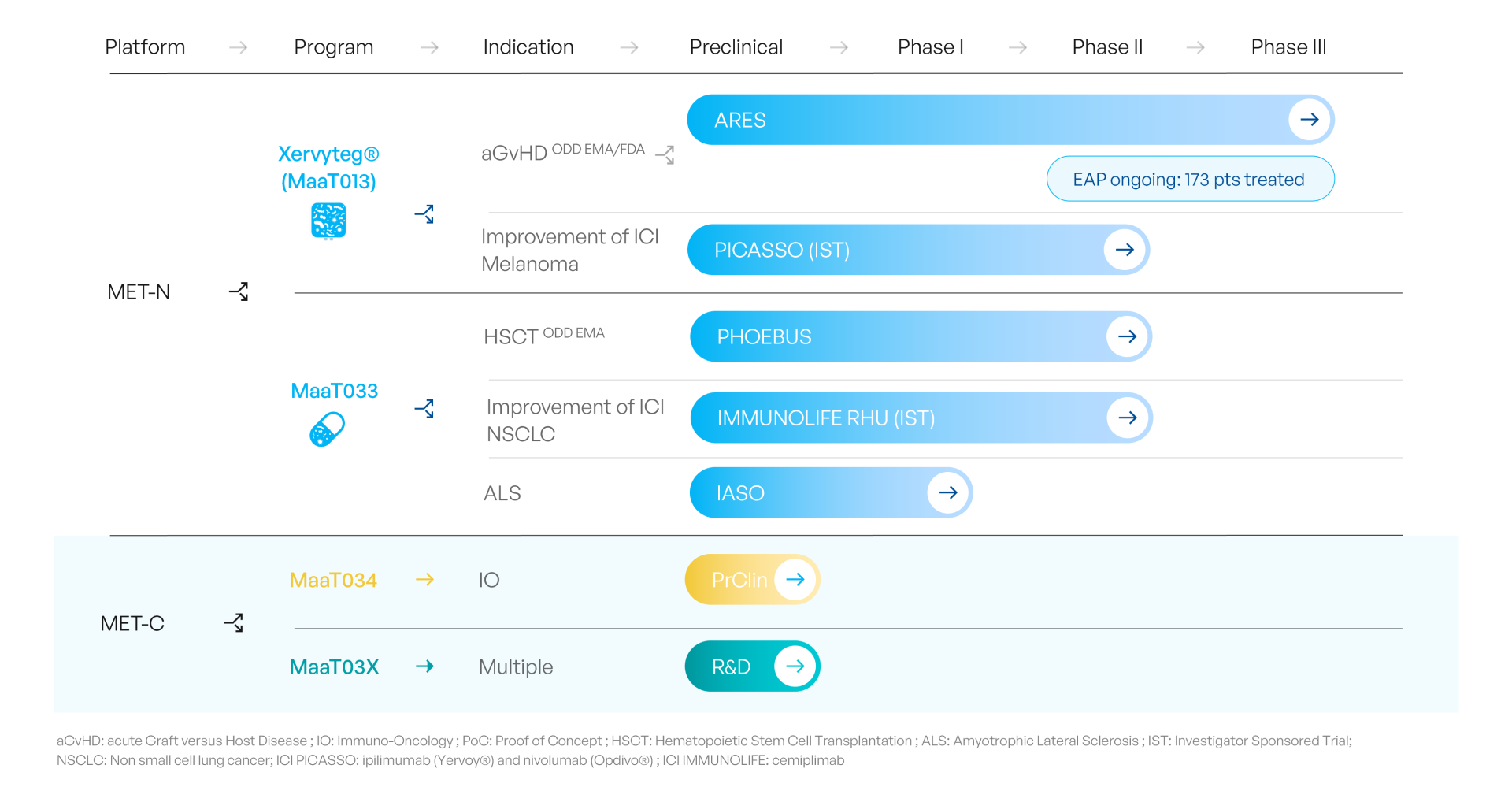
Restore in hemato-oncology:
MaaT Pharma’s therapies for the treatment of acute Graft-vs-host-Disease and to improve survival in patients receiving hematopoietic stem cell transplantation. This aims to restore healthy microbiome functions to correct the impact of stressors such as antibiotics, chemotherapies.
Restore & modulate in immuno-oncology:
MaaT Pharma’s therapies for the treatment of solid tumors aim to restore and modulate the microbiome to improve patient’s response to immune checkpoint inhibitors.
The standard first line therapy for treating aGvHD is the use of systemic steroids. If patients do not respond to steroids, they are considered Steroid Resistant (SR) and other agents can be administered such as ruxolitinib. Antibiotic and chemotherapy-derived loss of gut diversity is predictive of graft-vs-host-disease occurrence, as well as poor outcomes and lower survival in patients receiving allo-HCT. 2,3, 4,
Xervyteg® (MaaT013) is a donor-derived, standardized, high-richness, high-diversity microbiome ecosystem therapy, containing ButycoreTM , a group of bacterial species known to produce anti-inflammatory short-chain-fatty acids. Xervyteg® (MaaT013) has received Orphan Drug Designation from FDA and EMA.
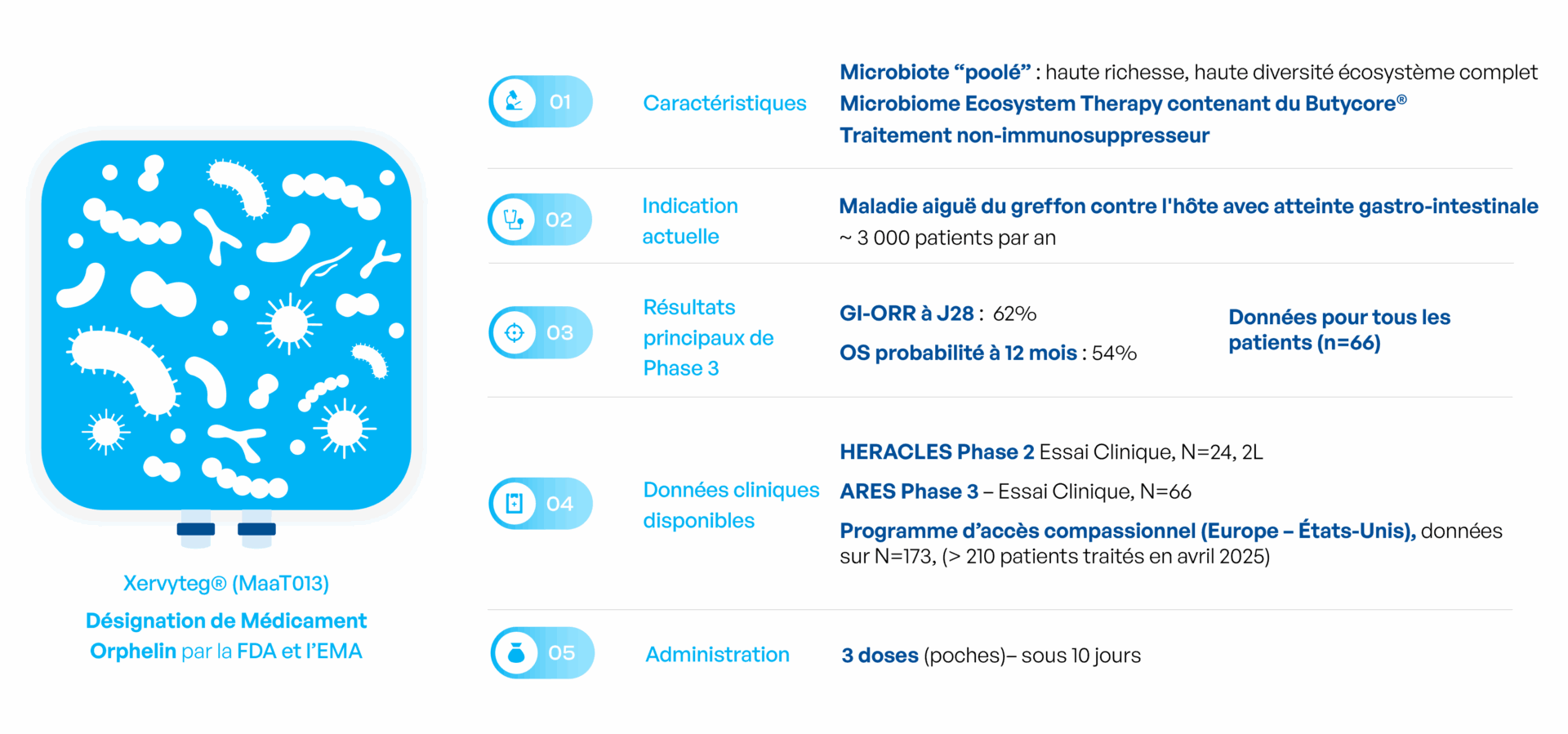
Xervyteg® (MaaT013) is a microbiota-based therapeutic currently under regulatory review in Europe for the treatment of gastrointestinal acute Graft-versus-Host Disease (GI-aGvHD). The therapy has been evaluated in the pivotal ARES clinical trial (NCT04769895), the first global pivotal study of a microbiome-based therapy in hemato-oncology.
The ARES trial enrolled in Europe 66 patients with GI-aGvHD refractory to both first-line steroid treatment and second-line ruxolitinib, the current standards of care.
patients treated to date
Key Clinical Milestones:
– First patient dosed in ARES pivotal clinical trial, confirming safety profile
– Positive Outcome Following DSMB Review for ongoing Pivotal study
– Positive Topline Results from the Pivotal Phase 3 ARES Study Evaluating MaaT013 in acute Graft-versus-Host Disease, showing high efficacy and favorable safety outcomes
– Positive Outcomes from Final DSMB Meeting for Pivotal Clinical Trial Evaluating MaaT013 in Acute Graft-versus-Host Disease, confirming positive benefit-risk profile
– Marketing Authorization Application submitted to the European Medicines Agency (EMA) in June 2025 for the treatment of GI-aGvHD
Allogeneic Hematopoietic Stem Cell Transplantation (allo-HCT) is a potentially life-saving procedure for people suffering from blood cancers such as acute myeloid leukemia.
Although potentially life-saving, allo-HCT can be associated with severe complications, such as severe infections, graft-vs-host-disease, neutropenia, contributing to a mortality rate of 34% after 1 year6 in patients over 50 years of age receiving allo-HCT.
patients receive an allo-HCT each year in the US, UK, France, Germany, Italy, Spain and Japan.
Antibiotic and chemotherapy-derived loss of gut diversity is predictive of mortality in allo-HCT patients, as well as graft-vs-host-disease occurrence and infection.2 3 4 7
MaaT033 is a an oral, donor-derived, standardized, high-richness, high-diversity microbiome ecosystem therapies, for ambulatory use, containing ButycoreTM , a group of bacterial species known to produce anti-inflammatory short-chain-fatty acid. MaaT033 has received Orphan Drug Designation from the EMA.

MaaT033’s proposed mode-of-action is immuno-modulatory:
Positive Phase 1b trial results
In June 2022, MaaT Pharma reported positive results for MaaT033 in Phase 1b clinical trial for patients with acute myeloid leukemia including a good microbiome engraftment and engraftment persistence, together with a satisfactory safety profile for its second drug-candidate.
Additional information:
Ongoing Phase 2b trial
In November 2023, MaaT Pharma announced that the first patient has been treated as part of its Phase 2b trial, called PHOEBUS, investigating the efficacy of MaaT033 in improving overall survival (OS) at 12 months for patients with blood cancer receiving allogenic Hematopoietic Stem Cell Transplantation. The trial is an international, multi-center, randomized, double-blind, placebo-control study (NCT05762211), which will be conducted in 59 clinical investigation sites and is expected to enroll 388 patients. It is to date the largest randomized controlled trial assessing a microbiome therapy in oncology.
Recent studies have has started to shed light on the role of the gut microbiome, linking abnormalities to diseases such as Amyotrophic lateral sclerosis (ALS[8]). The link between gut microbiota and ALS first emerged from preclinical evidence and then from clinical observations indicating a disease-modifying role for the gut microbiome.
To date, there is no effective treatment for ALS, a disease that leads to death within on average 3-5 years after diagnosis[9]. Since its inception, MaaT Pharma has been committed to restoring a microbiome symbiosis in life-threatening diseases with high unmet clinical needs. Growing evidence suggests that ALS patients show increased inflammation in the gut and changes in the composition of gut microbes, with low levels of beneficial bacteria.
This pipeline expansion to a new indication demonstrates the strong potential of MaaT033 to be used in acute or in chronic conditions as a standalone, adjunctive therapy.


1 Castilla-Llorente. C, et al. Prognostic factors and outcomes of severe gastrointestinal GVHD after allogeneic hematopoietic cell transplantation. Bone Marrow Transplant. 2014 49(7):966-71. doi:10.1038/bmt.2014.69
2 Peled JU, et al. Role of the intestinal mucosa in acute gastrointestinal GVHD. Blood. 2016 128(20):2395-2402. doi: 10.1182/blood-2016-06-716738.
Staffas A, et al. The intestinal microbiota in allogeneic hematopoietic cell transplant and graft-versus-host disease. Blood. 2017 129(8):927-933. doi: 10.1182/blood-2016-09-691394. Erratum in: Blood. 2017 Apr 13;129(15):2204
Mathewson ND, et al. Gut microbiome-derived metabolites modulate intestinal epithelial cell damage and mitigate graft-versus-host disease. Nat Immunol. 2016 505-513. doi: 10.1038/ni.3400. Epub 2016 Mar 21. Erratum in: Nat Immunol. 2016 Sep 20;17 (10 ):1235.
3 Taur Y, et al. The effects of intestinal tract bacterial diversity on mortality following allogeneic hematopoietic stem cell transplantation. Blood. 2014 124(7):1174-82. doi:10.1182/blood-2014-02-554725.
Jenq RR, et al. Regulation of intestinal inflammation by microbiota following allogeneic bone marrow transplantation. J Exp Med. 2012 903-11. doi: 10.1084/jem.20112408.
4 Jenq RR, et al. Intestinal Blautia Is Associated with Reduced Death from Graft-versus-Host Disease. Biol Blood Marrow Transplant. 2015 1373-83. doi:10.1016/j.bbmt.2015.04.016.
Peled JU, et al. Intestinal Microbiota and Relapse After Hematopoietic-Cell Transplantation. J Clin Oncol. 2017 1650-1659. doi:10.1200/JCO.2016.70.3348
5 Global Data GVHD Epidemiology Report, Jan 2020.
6 EBMT data 2021
7 Malard, F., Vekhoff, A., Lapusan, S. et al. Gut microbiota diversity after autologous fecal microbiota transfer in acute myeloid leukemia patients. Nat Commun 12, 3084 (2021). https://doi.org/10.1038/s41467-021-23376-6
[8] Rowin et al., 2017; Nicholson et al, 2021; Blacher et al, 2019, Mazzini et al, 2020
[9] https://tousensellescontrelasla.fr/la-sla-cest-quoi/
[10] Yishan Ye, Wenjing Hao, Florent Malard, Mohamad Mohty, Emerging novel therapies for steroid-refractory acute graft-versus-host disease: recent advances and future directions, Current Opinion in Immunology, Volume 96, 2025, https://doi.org/10.1016/j.coi.2025.102649.
[11] Emmanuelle Godefroy, Frédéric Altare, Deciphering and harnessing gut microbiota–associated immune regulation in acute graft-versus-host disease, Current Opinion in Immunology, Volume 97, 2025, https://doi.org/10.1016/j.coi.2025.102676.
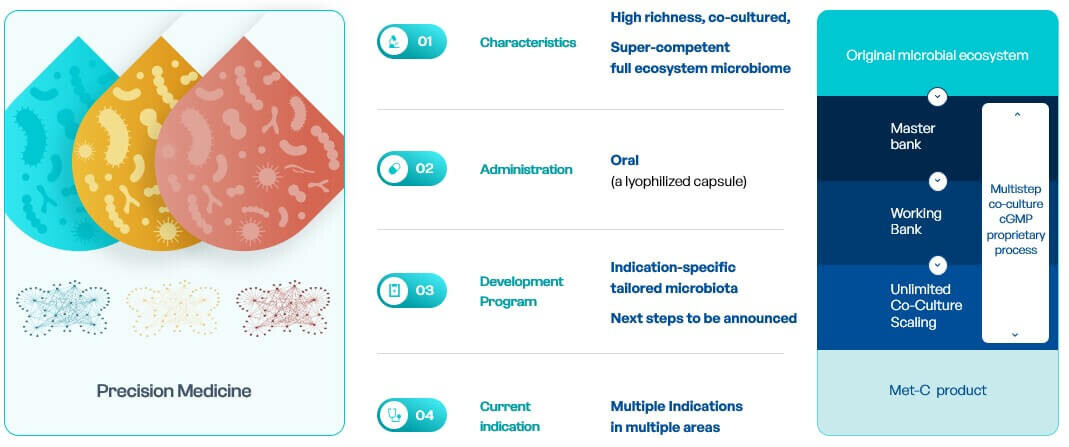
Immune checkpoint inhibitors (ICI) have proved a formidable therapeutic option for the treatment of solid tumors. They have notably improved survival in more than 15 oncology indications to date. However, not all patients respond to ICI. In some indications, it has been suggested that the gut microbiome plays a role in the response to ICI. Recent research has suggested that a patient’s gut microbiome diversity and richness, as well as its composition, impacts response rate to ICI and overall survival rate in multiple indications (melanoma, non-small cell lung cancer, renal cell carcinoma…).
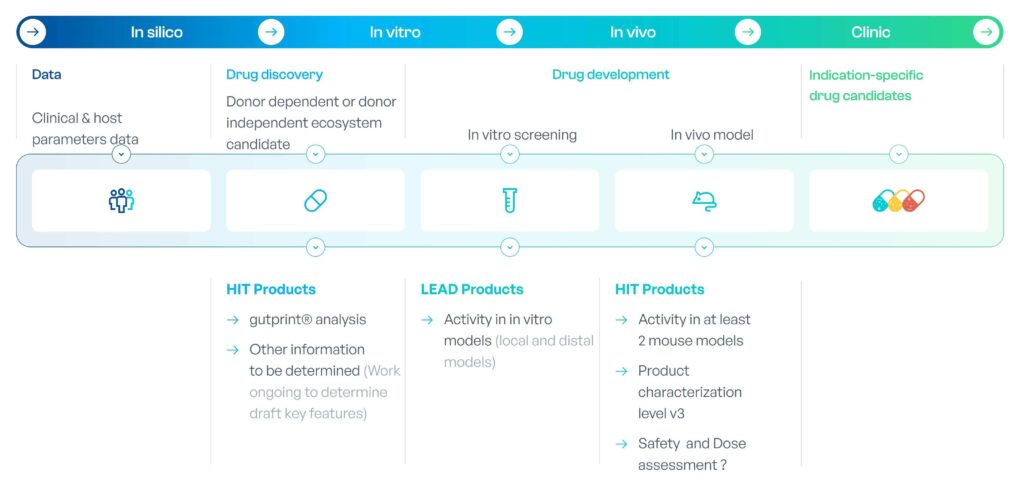
Using our gutPrint platform, we are designing indication-specific product candidates (MET-C) combining high richness and diversity and indication-specific functional bacterial networks aiming to improve response to ICI. Each product is designed based on AI-driven analysis of healthy subjects’ and patients’ data and manufactured using a unique, ground-breaking, patented, ecosystem co-culture technology allowing to replicate and leverage, at large industrial scale, the richness and diversity of native-based microbiome while tuning the resulting product to an indication-specific composition.
MaaT034, the first-in-class co-cultured product, is aiming at optimizing intestinal microbiome functions prior to and during immunotherapy in major oncology indications, and become a (neo)-adjuvant therapy across ICI therapies, leading to better survival in cancer patients. The objective of MaaT Pharma is to develop MaaT034 as tumor-agnostic adjunctive therapies to ICI (i.e. administration before and in combination with ICI whatever the type of cancer) leading to improved clinical outcomes and lower incidence of complications than using ICI alone.
To achieve this, MaaT Pharma will capitalize on MET-N products: an Investigator-Sponsored Trial (IST) is already ongoing in solid tumors with the PICASSO trial (metastatic melanoma, with Xervyteg®MaaT013) to evaluate the safety of MET-N products in combination with ICI and collect preliminary efficacy data assessing the impact of MET products on the immune system and anti-tumoral responses.

Sheri Simmons, Ph.D., is Acting Chief Scientific Officer at MaaT Pharma. Sheri brings extensive experience in biotechnology, particularly in the microbiome field, having held scientific leadership positions at Seres Therapeutics, Johnson & Johnson’s Microbiome Solutions team, and most recently at Seed Health, a leading probiotics company. In her role, she strengthens the Company’s scientific leadership, overseeing preclinical research, AI/data initiatives, and supporting efforts toward the Marketing Authorization Application of Xervyteg® in aGvHD.
Sheri holds a PhD in Biological Oceanography from the Massachusetts Institute of Technology (MIT) and completed an A.B. in Ecology & Evolutionary Biology at Princeton University, graduating summa cum laude and as a Phi Beta Kappa member, receiving one of six awards for the best senior thesis in the sciences. Sheri holds a PhD in Biological Oceanography from the Massachusetts Institute of Technology (MIT) and completed an A.B. in Ecology & Evolutionary Biology at Princeton University, graduating summa cum laude and as a Phi Beta Kappa member, receiving one of six awards for the best senior thesis in the sciences.
Hervé Affagard is the CEO and co-founder of MaaT Pharma. For the past 15 years, Hervé has been an intra/entrepreneur in the healthcare industry, after starting his career in IT in the steel industry. In late 2014, Hervé co-founded the company alongside Dr. Joël Doré, author of nearly 500 publications, and
one of the world’s most cited authors in the microbiome sphere today, after a professional career that spanned multiple industries. Hervé has led MaaT Pharma’s development from its early concept in 2013 and has been at the forefront of the development of the microbiome healthcare ecosystem in France and Europe. In January 2022, Hervé has been elected President of Allliance Promotion Microbiote, an organization founded in 2021 to support the microbiome sector development in France.
Engineer, MBA