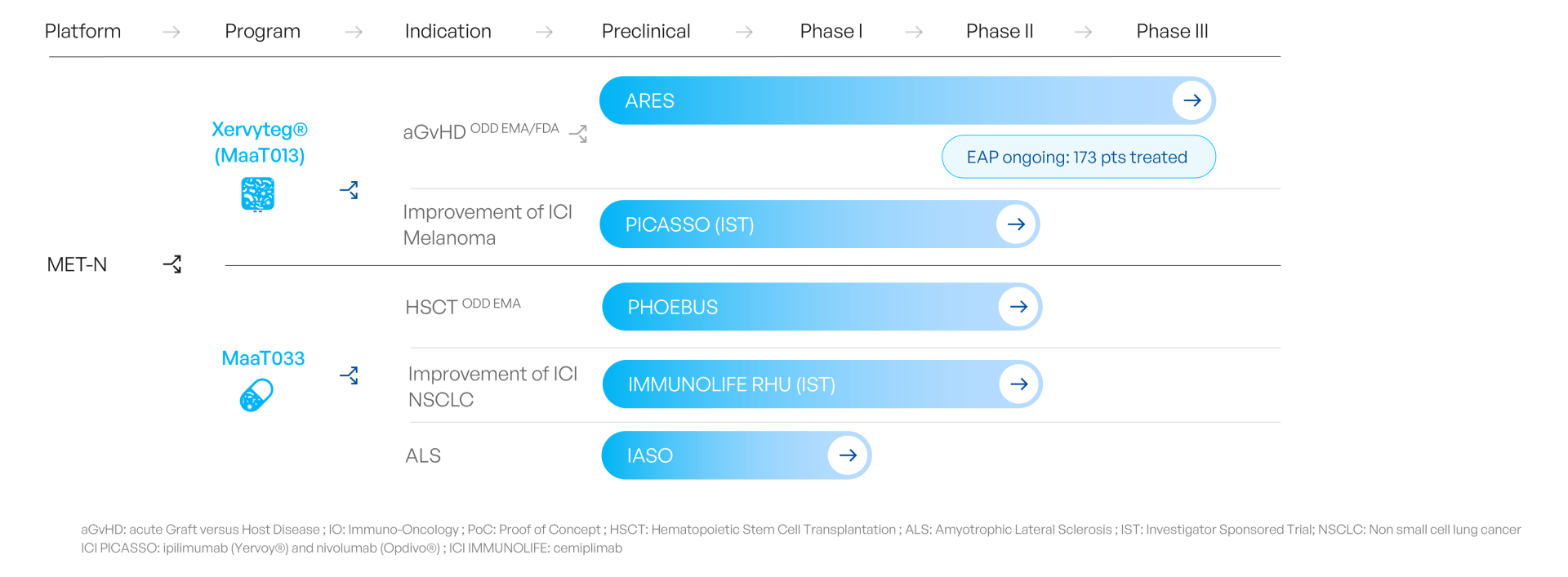
MaaT Pharma’s products, Microbiome Ecosystem Therapies (MET), are high-diversity, high-richness, indication-specific products aiming to leverage the full functional diversity of a large microbiome ecosystem.
MaaT Pharma developed one of the first global platform to develop and manufacture MET-N, native, donor-derived products. MET-N are intended as symbiosis-restoring therapies in indications where patients present an often iatrogenic, severe dysbiosis.
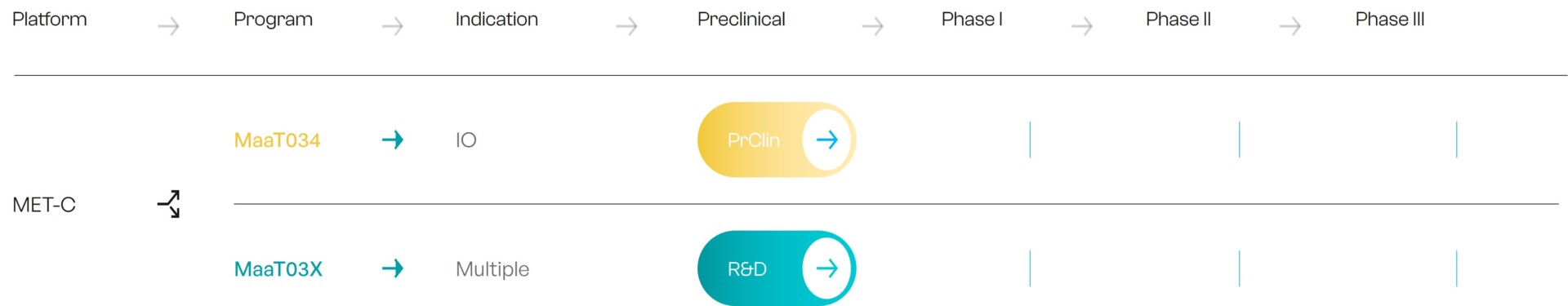
More recently, MaaT Pharma has leveraged the knowledge accumulated from native products to develop a new generation of products, MET-C, which can be rationally designed from patients-derived data and cultured at large scale. MET-C are intended as immune-modulating therapies in patients with solid tumors receiving immune checkpoint inhibitors.
MaaT Pharma has been a pioneer in establishing rigorous screening and industrial cGMP manufacturing processes to deliver donor-derived, standardized, high-richness, high-diversity MET.
True to the philosophy of METs, Xervyteg® (MaaT013) and MaaT033 not only contain a highly diverse, rich ecosystem, but are
also enriched, through a patented industrial process, in specific bacteria of interest, the ButycoreTM, selected for their ability to
produce anti-inflammatory metabolites such as Short Chain Fatty Acids.
While MaaT Pharma has established a solid proof of concept with native products, large indications will require large-scale manufacturing only accessible through culture technology.
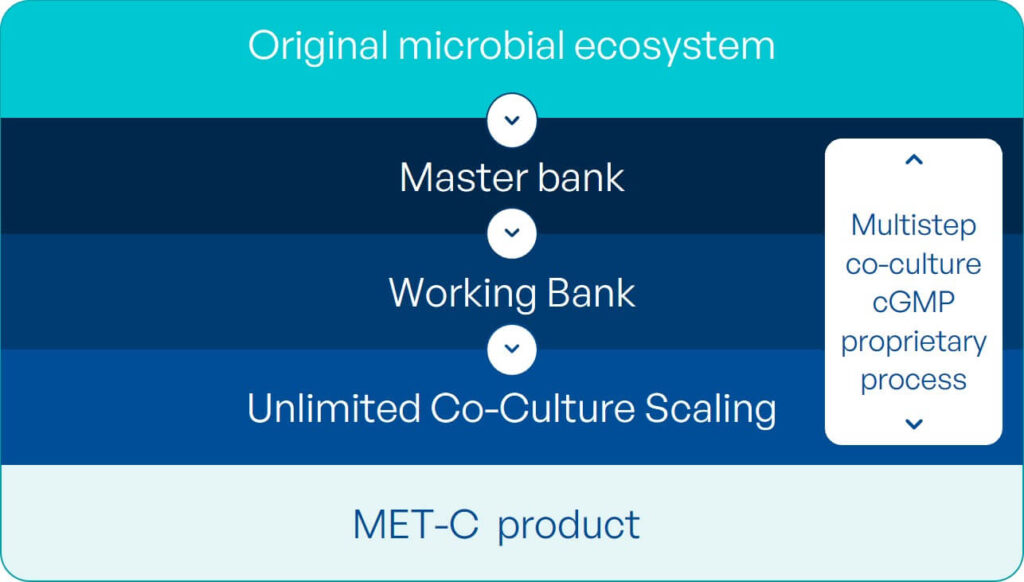
To address this need, MaaT Pharma has developed a unique, ground-breaking, patented co-culture technology allowing to replicate and leverage, at large industrial scale, the richness and diversity of native-based microbiome ecosystems while tuning the resulting product to indication-specific compositions.
MET-C candidates are based on AI-driven analysis of healthy subject’s and patient’s microbiome data.
As an example, MaaT03X (for the treatment of an undisclosed solid tumor) contains hundreds of co-cultured species, including both supportive and indication-specific functional networks.
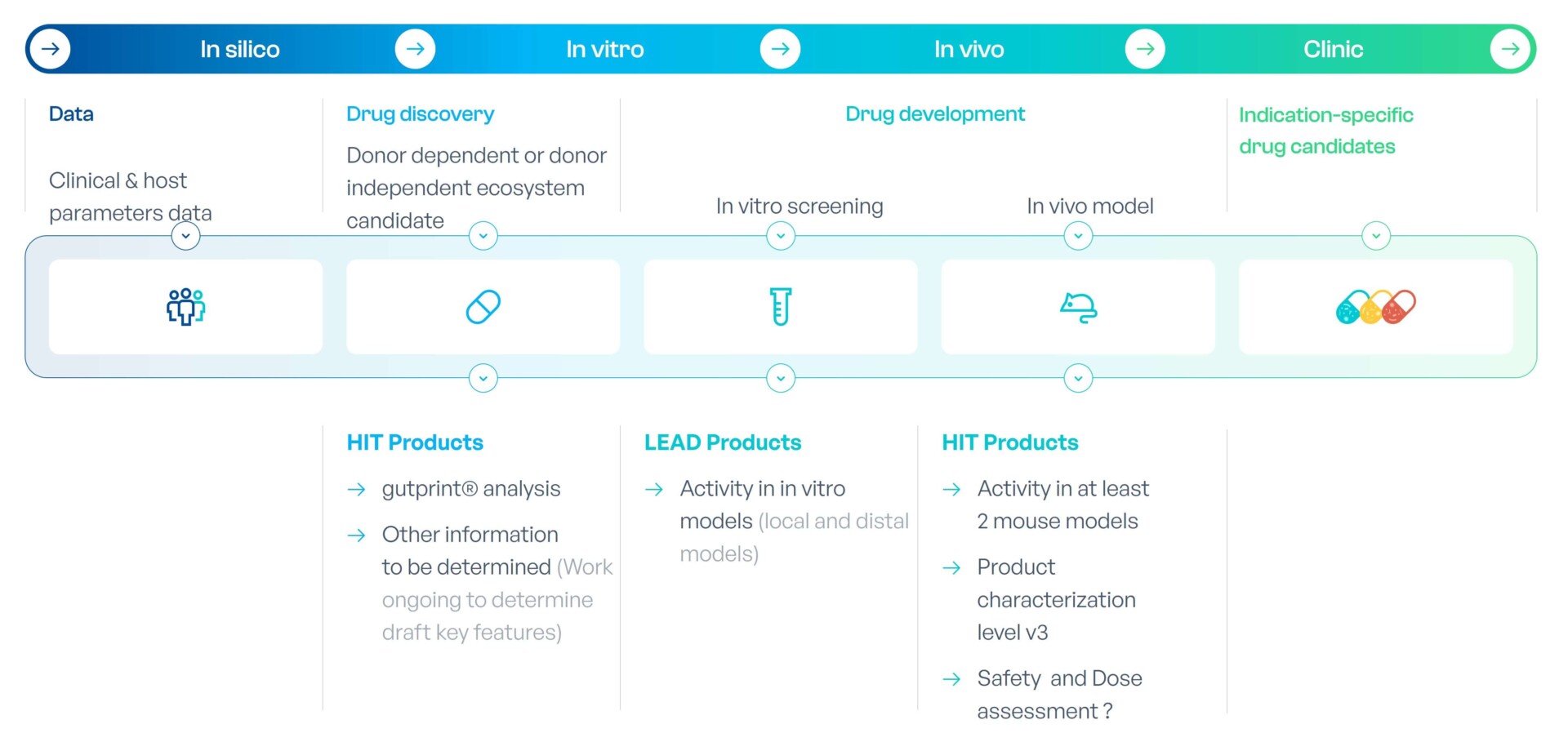
gutPrint® is the engine that drives the generation of new METs. It is MaaT Pharma’s AI-powered, proprietary, integrated drug discovery platform built to design, identify, monitor and validate disease-specific functional MET.
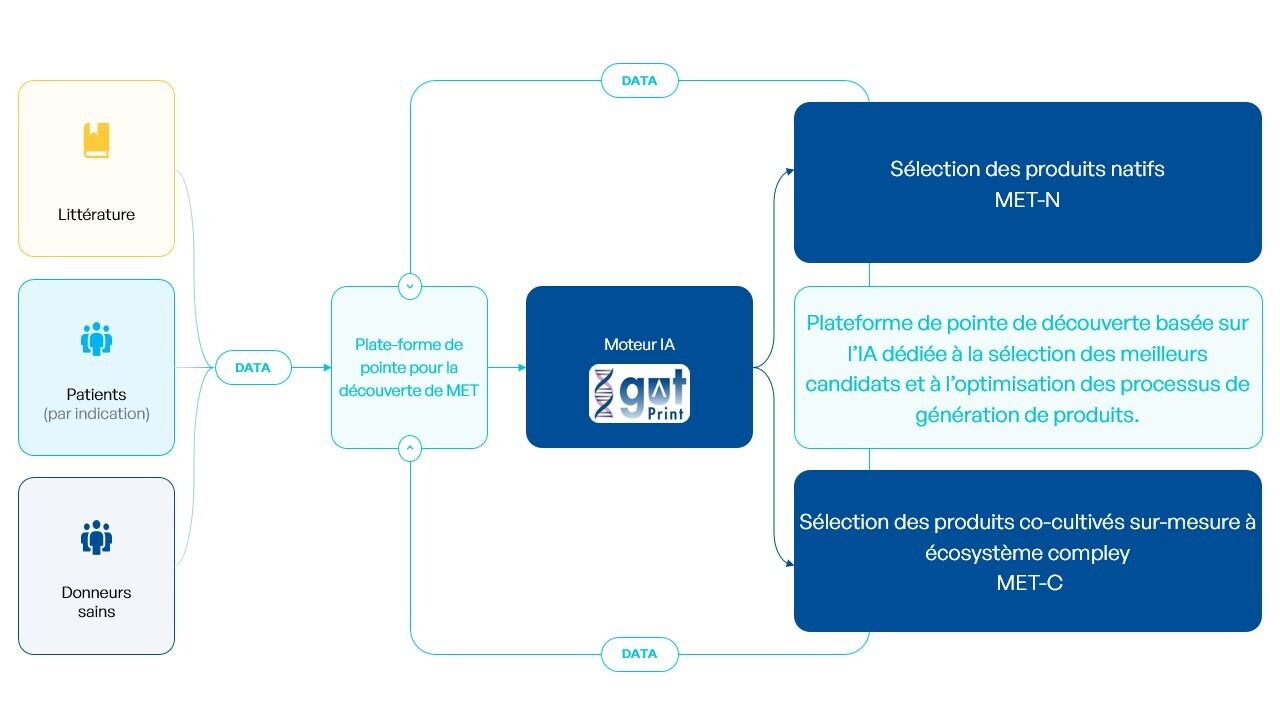
Metagenomic and biological data and insights from healthy donors, clinical studies as well as the literature and microbiome databases are fed into our platform to characterize supportive and functional microbial networks that are clinically relevant to each disease of interest, and design new MET to improve clinical outcomes. gutPrint® is also used to monitor drug development and manufacturing.
Each MET candidate is then validated using disease-appropriate in vitro assays and in vivo models of the disease leading to the final microbial composition of our drug candidate. Each step generates additional data that enrich the database and our understanding of the host/microbiome relationship.

MaaT Pharma has developed unique cGMP manufacturing and supply chain capacities to support the development of its products.
Partnering with Skyepharma, the Company has built, in record 12 months, Europe’s largest manufacturing facility entirely dedicated to microbiome-based drugs candidates.
Standardization is achieved with:
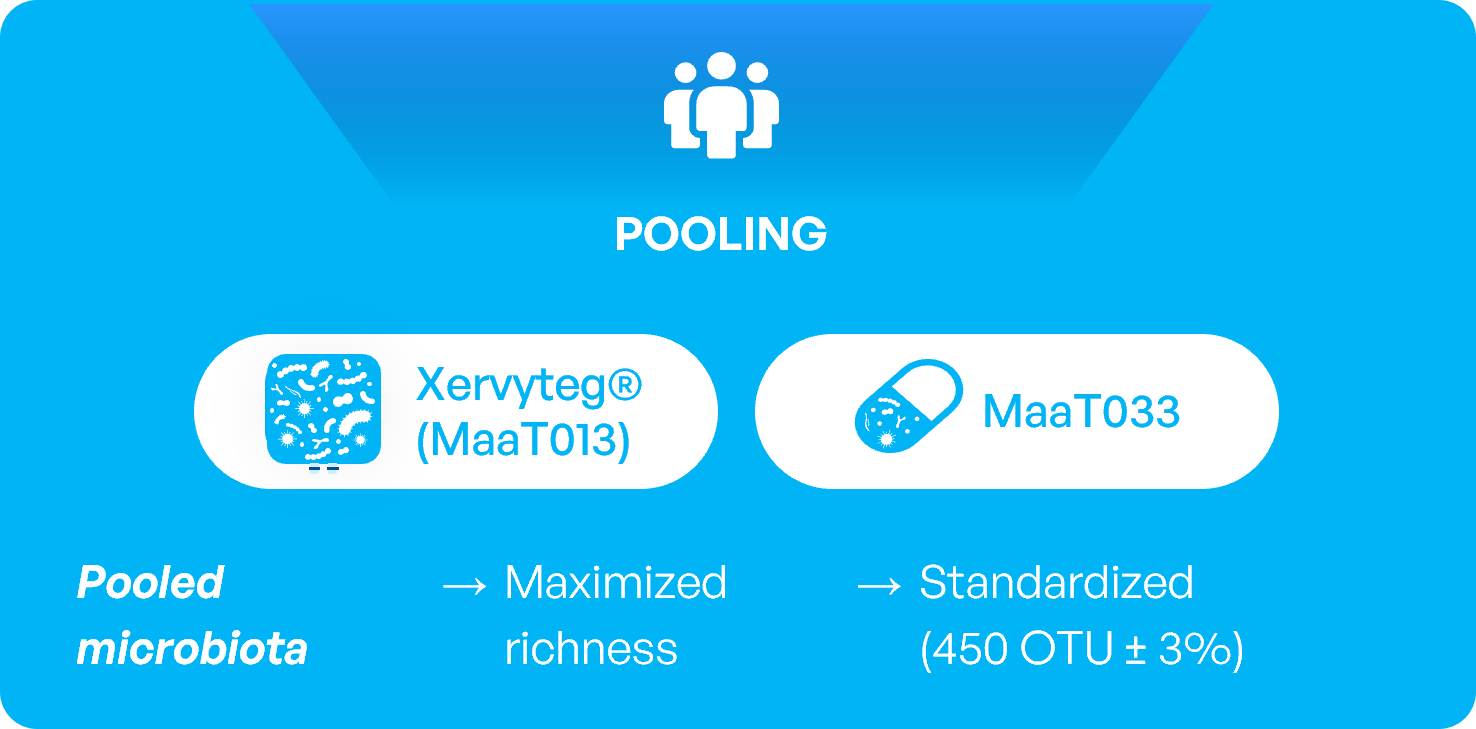
Recent paper published in Applied and Environmental Microbiology, titled “Reduction of product composition variability using pooled microbiome ecosystem therapy and consequence in two infectious murine models” highlighted compelling data supporting the superiority of pooling fecal microbiota from healthy donors over the single donor approach. For the single donor approach, results are variable and are all dependent from the donor while the pooling strategy allows for consistency and standardization of product activity.

”We pioneer cGMP process
to develop Microbiome
Ecosystem Therapies.
